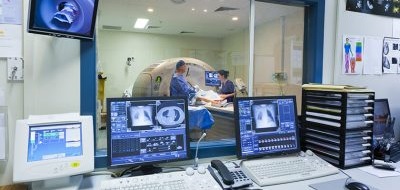
The Network collates Clinical Research Facility (CRF) capacity and capability information and works with stakeholders to promote CRFs as an integrated national platform for the delivery of high intensity experimental medicine studies to research funders.
If you are a research funder please visit our Industry page to submit information requests regarding potential studies or see our Clinical Research Facility page to learn about their unique features.
Work Package Activities
Database of CRF Capabilities
Signpost Industry Enquiries

WP2 Steering Group, March 2019
Working Groups
Work Package Groups comprise teams of expert individuals able to offer advice and guidance on their specialist areas, helping the UKCRF Network to achieve its objectives in supporting CRFs and potential Industry partners. For the Strategic Leadership Work Package, the Groups are:
Information Management and Technology Working Group
The Information Management and Technology (IM&T) Working Group shares ideas and expertise related to Information Systems Developments; emerging software technology, and Data and Information Management. Activities include considering cross-site IM&T developments, the use of commonly developed systems and making IT recommendations to CRF Senior Management.
Industry Liaison Working Group
Experimental Medicine Operations Manager at NIHR Cambridge Clinical Research Facility
Group Co- Chair: Colette Inkson
Innovation and Partnerships Lead at NIHR Manchester Clinical Research Facility
Details about the Working Groups can be found on the UKCRF Network Portal.
If you would like to join any of our Work Package Groups please contact us.

Work Package resources
The UKCRF Network recommend the use of the NIHR Industry Costing Template for commercial studies and the UKCRF Network Study Intensity Tool for costing non-commercial studies run in CRFs.