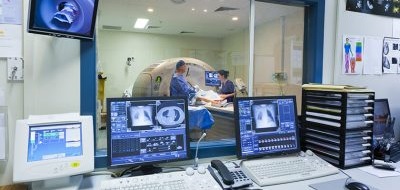
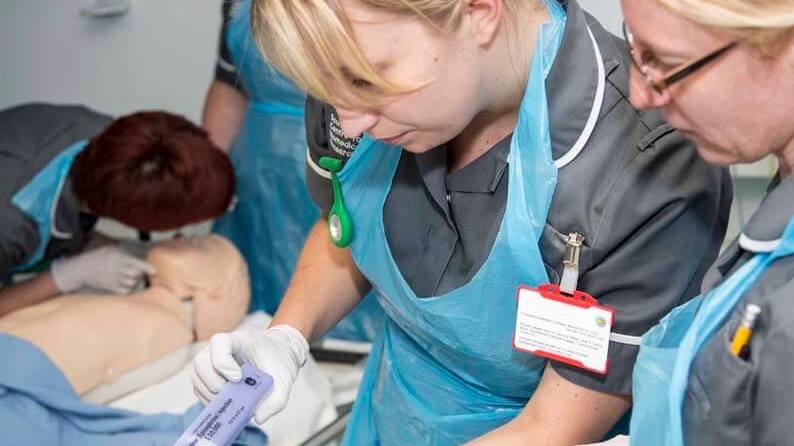
Clinical Research Facilities (CRFs) for Experimental Medicine are dedicated facilities, where specialist clinical research and support staff from universities and NHS Trusts work together on patient-orientated commercial and non-commercial experimental medicine studies. We have over 50 CRFs within our Network, with over 110 individual units across the UK and Ireland.

The UKCRF Network collaborates with Clinical Research Facilities and other experimental medicine infrastructure across the UK and Ireland to develop, share and implement excellence in operational practice to ensure the efficient and effective delivery of studies, and drive forward initiatives that improve quality of patient experience.
About Us
Read MoreDeveloping Best Practice
Read MoreThe UKCRF Network develops guidelines, training materials, spreadsheets and check lists to support CRF operational excellence and workforce development. Our tools and guidelines can be used by all clinical research staff across the UK and Ireland.
9 - 10 July
UKCRF Network Annual Conference
Two Islands, Many Voices – Futurescaping Trans-National Clinical Research
Trinity college, Dublin, hosted by Ireland's Clinical Research Facilities.