October 2020
The COVID-19 pandemic has required an urgent response from both clinicians and researchers across the UK to clinically support patients and ensure clinical research studies are set-up rapidly to find effective diagnostics, treatments and vaccines.
Nationally, Clinical Research Facilities (CRF) have joined forces with their local Research and Innovation Departments and Universities to support both clinical and research requirements during the pandemic.
NIHR Cambridge CRF provides one example of the hard work and contributions made by CRFs during this challenging time:

.

Written by
Caroline McMahon (pictured left)
Patient & Public Involvement Lead, NIHR Cambridge CRF
Caroline Saunders (pictured right)
Director of Clinical Operations, NIHR Cambridge CRF
1. We established and led teams from across the campus to deliver Complex COVID-19 research trials across the Trust in record time:
COVIDx (SAMBA) Study – investigating a new COVID-19 test. We validated the rapid point-of-care test using Samba II machines, adapted from kit originally designed for HIV testing in developing countries by a University of Cambridge spin out called Diagnostics for the Real World. The test was trialled on an NHS COVID triage admissions ward and was adopted by the Trust in just 4 weeks. The device delivers results in 90 minutes enabling timely placement and discharge of patients (learn more).
Oxford Vaccine Trial – over 700 Health Care Workers recruited and 300 dosed in 3 weeks to the first UK vaccine trial (in partnership with Cambridge University Hospitals, Royal Papworth Hospital and Cambridgeshire and Peterborough NHS Foundation Trust). We had the first BAME participant & the only UK mental health trust to be involved (learn more).
.
TACTIC platform trial – testing whether re-purposing existing drugs that target the body’s immune response, can prevent severe organ failure or death from COVID-19. We treated 12 patients on CUH wards (learn more).

Staff supporting the Oxford COVID Vaccination Study (left); Collecting COVID-19 samples and using the point-of-care Samba machines (right)
2. We continued treating patients on over 80 ‘never stop’ clinical trials
Patients recruited to clinical trials have continued to receive potentially life-saving experimental drugs throughout the COVID pandemic. This includes patients with advanced cancer diagnoses.

Professor Farooqi receiving a shipment of 20,000 face-masks,
thanks to their collaboration with colleagues in China
3. We supported key Hospital services:
Our Clinical Investigation Ward hosted Pleural Services so that interventions could continue in this patient population.
Undertook day-case standard-of-care minor procedures for cancer patients (relocated from Addenbrooke’s Treatment Centre) and continued with 2 week cancer wait diagnostic services using non-endoscopic pill-on-a-string cytosponge device developed in Cambridge and tested in multiple studies in the CRF.
Delivered standard-of-care treatment to Oncology Day Unit trial patients where capacity within these service areas was compromised.
Delivered respiratory patient management training to over 850 CUH staff in 5 weeks.
.
Established combined standard of care and research follow-up clinics for patients admitted with COVID-19.
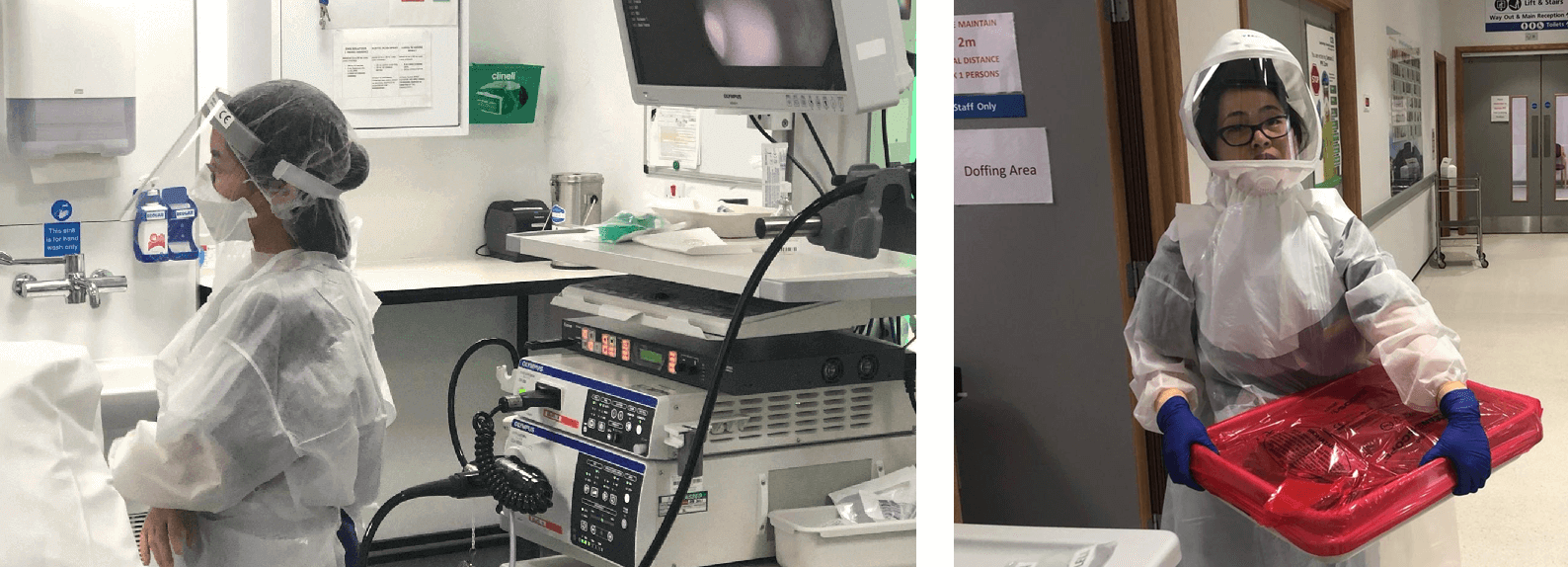
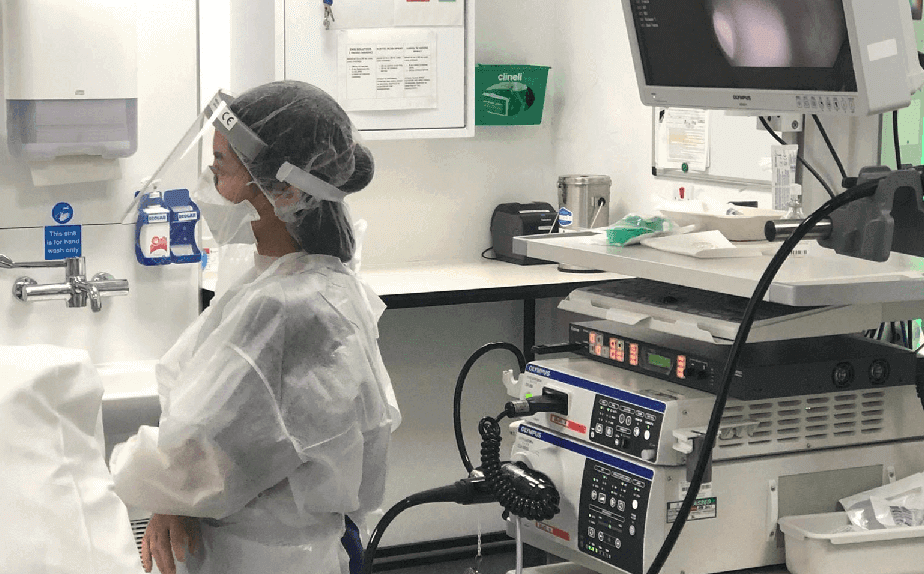
Demonstrating full PPE for Endoscopy
4. We worked in partnership with Occupational Health to establish COVID staff testing:
Walk-in on-site staff testing stations (PODs) were established to deliver mass testing for NHS & University staff on campus. This initiative enabled asymptomatic transmission by staff to be tracked and managed. Over >11,000 CUH staff have been tested to date (October 2020) (learn more).
.
Staff serology testing – we delivered mass staff testing in partnership with Occupational Health and through this recruited over 5,900 to NIHR COVID-BioResource. Samples will be used to study the pathophysiology of the disease over time, including infection, recovery and mental-health outcomes (learn more).

Outside the COVID staff testing PODs in all weathers
Thank you to everyone involved
The whole team at NIHR Cambridge CRF showed dedication, expertise and ability to turn their hand to everything that has been asked of them, we are so proud. Thank you everyone!