The UKCRF Research Nurses and Practitioners Group have developed a bedspace checklist to ensure the safety of participants in early phase clinical trials.
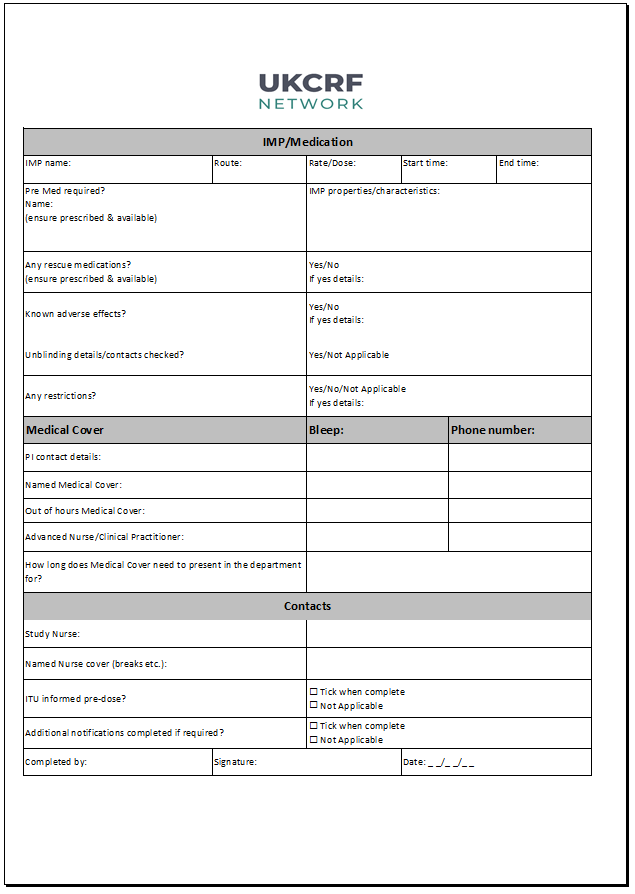
The Dosing Day Final Checklist (formally the early phase bedspace checklist) has been designed to provide a framework for final safety checks. The checklist ensures all safety governance procedures are in place prior to exposing participants to investigational medicinal products (IMP).
The checklist was piloted at six CRFs (Leicester, Newcastle, Exeter, Lancashire, Sheffield and Cambridge) and is now available for download from the UKCRF Portal.
WHAT DOES THE Early phase bedspace CHECKLIST INCLUDE?
- Room checks (e.g. emergency buzzer, suction and oxygen)
- Participant medical history and observations
- Emergency checks (crash trolley and anaphylaxis)
- IMP details (e.g. dose, pre-medication, known adverse effects etc.)
- PI and medical cover contact details

HOW CAN THE DOCUMENT BE ACCESSED?
The new checklist can be downloaded from the UKCRF Network Portal. If you are not already a user, complete the request form on the Portal page to gain access.
Acknowledgements
Stewart Fuller, Lead Nurse at NIHR Cambridge CRF led the sub-group to create this checklist with support from members of the Research Nurses and Practitioners Group.
The checklist was piloted at six CRFs; Leicester, Newcastle, Exeter, Lancashire, Sheffield and Cambridge.




